LA JOLLA, CA—Scientists at La Jolla Institute for Immunology (LJI) have shed light on how an unusual population of T cells may drive harmful inflammation in people with ulcerative colitis, an autoimmune disease that causes damage to the large intestine.
Their new study, published recently in Nature Immunology, is the first to show a link between “stem-like” T cells and ulcerative colitis in human patients.
“We’ve found a population of T cells that may be important in disease—and may contribute to relapse in patients with ulcerative colitis,” says LJI William K. Bowes Distinguished Professor Pandurangan Vijayanand, M.D., Ph.D., who co-led the study with LJI Professor Mitchell Kronenberg, Ph.D.
The researchers hope to one day target this T cell population via drug therapy. “These cells may be a very important target for treating ulcerative colitis and perhaps other autoimmune diseases,” says Kronenberg.
Investigating the origins of ulcerative colitis

T cells normally help the body fight pathogens, such as viruses and bacteria. In autoimmune disease, the body’s T cells mistakenly begin attacking a person’s healthy tissue. For patients with ulcerative colitis, T cells cause chronic inflammation in the large intestine, leading to devastating tissue damage and potentially life-threatening complications. Around half of patients respond to therapeutics, but disease relapse is very common.
For the new study, the LJI team combined their expertise in immunology, cell biology, and genomics to answer an important question: Where do all these harmful T cells come from?
Typical T cells engage with their targets (such as viral antigens) and eventually become dysfunctional—or even die—upon repeated stimulation. The body wants T cells to diminish their responses or die after they do their jobs. That ensures the T cells won’t hang around and trigger any off-target inflammation.
But stem-like T cells have tapped into a fountain of youth. “These cells can self-renew and can give rise to more stem-like cells, but they can also give rise to the really pathogenic cells,” says Kronenberg.
One identifying feature of some stem cells and stem-like T cells is a gene called TCF1, which acts in the nucleus to control the expression of many other genes. In previous studies, researchers had discovered that these stem-like T cells are common in animal models of autoimmune diseases, including diabetes and ulcerative colitis. When researchers examined the genes expressed by these stem-like T cells, they found the TCF1 gene to be a hallmark that distinguished these cells from other types of T cells.
A closer look at human patients

The LJI researchers needed to know more about these stem-like T cells, and they needed to know if these cells might be responsible for sustaining T cell activation in a chronic disease, such as ulcerative colitis in human patients.
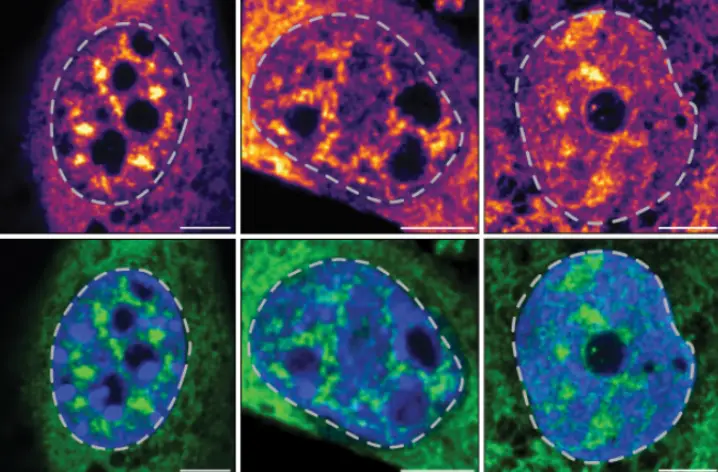
The LJI team analyzed colon tissue samples from human patients with ulcerative colitis. They looked closely at the individual T cell transcriptomes from thousands of cells, which told them which genes were active in T cells. This allowed the scientists to identify different T cell subsets present in diseased tissue. Patients with ulcerative colitis had a large stem-like T cell population in the large intestine. These cells were especially common in inflamed regions of the large intestine.
That correlative finding didn’t necessarily mean stem-like cells were driving the disease. To find out if they were the culprits, the researchers turned to animal models and looked at the genomes of T cells in the intestine of mice with colitis. They found the stem-like cells were likely the progenitors—or the source—of several types of pathogenic T cells.
The researchers also showed that they could trigger ulcerative colitis by transferring the stem-like T cells repeatedly into sets of healthy mice. The cells’ ability to sustain pathogenic potential illustrates their “stem-like” function.
The researchers then modified some stem-like T cells to be much less “stem-like” by removing the TCF1 gene. When they transferred these cells into mouse models of ulcerative colitis, the mice showed a reduction in pathogenic T cells.
The mouse data reinforced the idea that these stem-like cells may be responsible for fueling ulcerative colitis—and triggering relapses—in human patients as well.
“In a mouse model of colitis, we could show that stem-like T cells are required to sustain disease,” says Kronenberg. “These cells could be the thing that’s continuously feeding the fire under conditions of repeated antigen stimulation.”
The researchers emphasize that the current findings are basic scientific discoveries. “They need extensive validation before they can lead to therapeutics in humans,” says Vijayanand.
Vijayanand is especially interested in studying stem-like T cells specifically in ulcerative colitis patients who relapse. At the same time, he’s leading research to figure out how to eventually target these cells.
Kronenberg hopes the research can lead to new ulcerative colitis therapies that help more patients and reduce the risk of relapse. He says the new understanding of stem-like T cells may also help shed light on the origins of other autoimmune diseases.
Authors of the study, “Stem-like T cells are associated with the pathogenesis of ulcerative 1 colitis in humans,” included co-first authors Yingcong Li and Ciro Ramírez-Suástegui, as well as Richard Harris, Francisco Emmanuel Castañeda-Castro, Gabriel Ascui, Tamara Pérez-Jeldres, Alejandro Diaz, Carla Morong, Daniel A. Giles, Jiani Chai, Gregory Seumois, Tilman Sanchez-Elsner, and Fraser Cummings.
This study was supported by the De Laszlo Foundation, the William K. Bowes Jr Foundation, La Jolla Institute for Immunology (LJI) / Kyowa Kirin Inc. (KKUS) Interactive Fund., and the National Institutes of Health (grants DK46763 and F32 AI 466 140581).
DOI: 10.1038/s41590-024-01860-7
###